HSP40, YDJ1 Antibody Paper Review

HSP40, YDJ1 Antibody Paper Review

Copyright © StressMarq Biosciences Inc 2018-2019 All rights reserved
Our Mouse Anti-Yeast HSP40, YDJ1 Antibody was recently cited in a paper published in PLOS Genetics. The paper focuses the role of co-chaperones involved in ribonucleotide reductase (RNR) activity. RNR is an enzyme that converts ribonucleotides (NTPs) to deoxyribonucleotides (dNTPs). This is essential for DNA synthesis and RNR is an attractive target for anti-cancer therapies, since inhibiting RNR can promote apoptosis.
Molecular chaperones are proteins that assist in folding/unfolding or assembly/disassembly of other proteins, known as “clients.” Heat shock protein 70 (Hsp70) is one such chaperone. It is used extensively by unstable oncoproteins and is therefore highly expressed in certain types of cancer.
Hsp70 is a chaperone of RNR, which has other co-chaperones. This paper investigates which co-chaperones are involved with the RNR complex, which produces dNTPs to repair DNA that has been damaged. Inhibiting said co-chaperones could prevent RNR from repairing DNA and the cell cycle from progressing, which is an ideal outcome for cancer cells.
Hydroxyurea (HU) is a small molecule that inhibits RNR by reducing a tyrosyl radical in R2, an RNR subunit. It is used to treat cancer and sickle-cell disease, and stalls the DNA replication fork to generate single-stranded DNA.
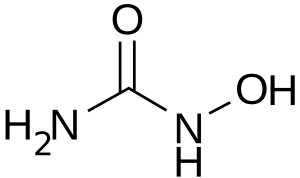
Hydroxyurea Chemical Structure. CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=657995
When Hsp70 or Hsp90 are inhibited, the RNR complex is destabilized and HU and other RNR inhibitors can have a greater effect. To find other targets that can increase the effect of RNR inhibitors such as HU, researchers at UNC Charlotte screened 28 yeast co-chaperone knockout strains for sensitivity to HU. If yeast without a certain co-chaperone showed increased sensitivity to HU, that co-chaperone was likely involved in RNR’s response to DNA damage. Yeast cells lacking Ydj1, a HSP40, were killed at lower concentrations of HU and were therefore more sensitive to HU than wild-type and other mutant yeast strains.
Researchers tested the role of Ydj1 in the DNA damage response pathway by performing a β-galactosidase activity assay and comparing the responses of wild-type and ydj1Δ cells to RNR3 promoter-lacZ, a DNA damage response-promoter. RNR3 is a gene expressed in response to DNA damage. As expected, cells lacking ydj1 had impaired RNR3 transcription, suggesting the DNA damage response was compromised. Therefore, Ydj1 is required for the DNA damage response and associated resistance to HU-induced cell death.
Researchers also found that ydj1Δ cells had decreased RNR2 and RNR4 transcription (and therefore decreased levels of RNR subunits Rnr2 and Rnr4). Rnr2 was significantly destabilized in ydj1Δ cells treated with HU. Ydj1 was found to interact with Rnr2 in yeast and its human equivalent, HDJ2, was found to interact with R2B, a subunit of RNR.
Treating HEK293 cells with 116-9e, a small molecule that blocks Hsp40-Hsp70 binding, disrupted the R2B-Hsp70 interaction. Hsp40s transport clients to Hsp70s for folding, so disrupting the interaction between HDJ2 and Hsp70 could be prevent cells from resisting the effects of HU through RNR-enabled DNA repair. 116-9e and HU had a synergistic effect inhibiting growth in HAP1 cells, which suggests a two-pronged approach of HDJ2 and RNR inhibitors may present a promising cancer treatment option.
The following was submitted from Nitika, a PhD student in the Truman Lab at UNC Charlotte:
“Hsp70 is a well-conserved molecular chaperone involved in the folding, stabilization, and eventual degradation of many “client” proteins. Hsp70 is regulated by a suite of co-chaperone molecules that assist in Hsp70-client interaction and stimulate the intrinsic ATPase activity of Hsp70. While previous studies have shown the anticancer target ribonucleotide reductase (RNR) is a client of Hsp70, the regulatory cochaperones involved remain to be determined. To identify co-chaperone(s) involved in RNR activity, 28 yeast co-chaperone knockout mutants were screened for sensitivity to the RNR-perturbing agent Hydroxyurea. Ydj1, an important cytoplasmic Hsp70 co-chaperone was identified to be required for growth on HU. Ydj1 bound the RNR subunit Rnr2 and cells lacking Ydj1 showed a destabilized RNR complex. Suggesting broad conservation from yeast to human, HDJ2 binds R2B and regulates RNR stability in human cells. Perturbation of the Ssa1-Ydj1 interaction through mutation or Hsp70-HDJ2 via the small molecule 116-9e compromised RNR function, suggesting chaperone dependence of this novel role. Mammalian cells lacking HDJ2 were significantly more sensitive to RNR inhibiting drugs such as hydroxyurea, gemcitabine and triapine. Taken together, this work suggests a novel anticancer strategy-inhibition of RNR by targeting Hsp70 co-chaperone function.
We are really thankful to StressMarq for generating antibodies for Ydj1. We used this antibody for Immunoprecipitation assay followed by western blot to demonstrate that Ydj1 interacts with Rnr2.
This work was recently published in PLOS Genetics and StressMarq antibody really helped us in our experiments. Apart from this, we work on other chaperones such as Hsp110 and I have used the polyclonal Hsp110 antibody. These are both excellent antibodies.”
 Loading ....
Loading ....
